Electric Transcription Discs
These discs are one of the most fragile formats and most at-risk of being lost due to deterioration, poor storage or mishandling. Transcription discs should be nearly, if not the top priority for preservation in many collections. If you have any of these discs in your archive or personal audio collection, you should be thinking about transfering them as they are highly unstable and fragile.
Electric transcription discs can also be referred to as
- Transcription discs (for short)
- ET discs (for short)
- Lacquer discs
- Instantaneous discs
- Nitrocellulose discs
- Cellulose nitrate discs
- Acetate discs (a misnomer)
In general, we will refer to these discs as "transcription discs" or "lacquer discs" below. Anything which is particularly important will be highlighted in orange - you can read just the orange text to get a quick and basic understanding.
Transcription discs blanks were manufactured under some of the following brand names (not an exhaustive list):
- audiodisc (Audio Devices)
- Duodisc
- Federal Perma-Disk or Perma Disk
- Howard Recording Disc
- Melodisc
- National Hollywood Recording Disc
- Philco
- Presto
- RCA De Luxe Recording Disc
- Recordio
- Recordisc
- Silvertone
- Soundcraft
- Tempo-Tone
- Tru-Test Recording
- Victor Home Recording Record Blank
- Voxite
- Wilcox-Gay
Transcription discs were manufactured with different degrees of quality for professional or home use.
What follows below is a detailed discussion on the following aspects of electric transcription discs:
- Important do's and don'ts
- Brief background
- Which discs to preserve
- Construction and materials
- Handling
- Cleaning
- Stylus selection
- Playback
- Storage
- Sleeve recommendations
Transcription discs are often mistaken for commercial recordings like 78s (shellac) or LPs (vinyl) because their appearance can be very similar to the untrained eye. However, the grooves are much softer than commercial shellac or vinyl discs and are easily damaged and worn. Also, transcription discs cannot be cleaned in the same way as vinyl or shellac discs.
DO NOT HANDLE WITHOUT GLOVES. Finger oil is acidic and will cause palmitic and stearic acid to form in the location of the finger print. Over time, the finger print will be literally etched into the disc laminate. The finger print will be both visible and audible, even after thoroughly cleaning the disc.
DO NOT PLAY these discs unnecessarily. Each play will further wear the grooves and deteriorate the sound. Changes in sound will easily be audible after a half-dozen plays, and changes can be measured in even fewer plays.
DO NOT USE LP STYLI when playing these discs. The discs may look like a modern LP, but at the microscopic level the grooves are 2-3x wider than LP grooves. An LP stylus will be too small, and will not fit the grooves properly - it will "rattle around" in the large grooves and damage the groove walls like a chisel.
DO NOT PLAY DISCS "WET". There is some folklore that suggests that quieter transfers can be achieved by wetting the surface of the disc with water or other lubricant. DO NOT DO THIS. The nitrocellulose laminate is highly hygroscopic (ie. it absorbs water) and will swell and possibly delaminate when exposed to water for a prolonged period of time. If a lubricant is used, it will need to be removed afterwards and that will require more water exposure. The risks far outweight any possible benefits. Only play these discs dry and without any lubricants or fluids.
DO NOT CLEAN these discs unless you have the knowledge, experience, and tools. Special chemistry, cleaning fluids, and equipment are needed to remove contamination as well as remove a white waxy substance or dust that forms on these discs with age called Palmitic Acid. Get trained or enlist a professional who knows how to clean these fragile discs.
DO NOT CLEAN SHELLAC OR VINYL DISCS with the same cleaning solutions as for transcription discs.
DO NOT CLEAN DISCS WITH DELAMINATION, BUBBLED OR CRACKED LAMINATE. Any exposure to water will rapidly exacerbate the delamination or cause delamination. At most, spot clean areas of contamination that are not delaminated, bubbled, or cracked.
DO NOT CLEAN DISCS ON A FIBER SUBSTRATE in the same way as glass or metal substrates. The fiber will expand and damage the laminate. At most, a moist but not wet brush can be used, and the moisture should be rapidly removed with a vacuum-style record cleaning machine like the Kieth Monks. Consider only spot cleaning areas of contamination that are not delaminated or cracked.
DO MAKE A TRANSFER OF DISCS BEFORE CLEANING if the laminate is delaminating, peeling, bubbled or cracked. Cleaning should, in general, be avoided on badly deteriorating discs unless the disc is also suffering from palmitic acid deposits. Even then, only a very light cleaning or spot cleaning at best using a moist brush, very light pressure, and a vacuum-style record cleaning machine.
DO STORE IN A DRY PLACE. The discs will rapidly deteriorate in the presence of moisture. Basements, garages, and unsealed and uninsulated storage sheds are not good places to store these discs.
DO STORE IN A COOL PLACE. The discs will rapidly deteriorate when exposed to heat, and even more so when the temperature fluctuates more than a few degrees. For example, the attic is not a good place to store these discs.
Brief Background on Transcription Discs
The electric transcription disc was predominantly used from the late 1920s (the dawn of electric recording) through the early 1960s as magnetic tape became the dominant recording format.
As a means to record audio, the electric transcription disc pre-dates wire and magnetic tape recordings. All commercial disc recordings (78s, LPs, 45s) were recorded on electric transcription discs at some point. Transcription discs found their way into many recording processes, including:
- Commercial recordings
- Radio programs and test pressings
- Office dictations
- Home recording
- Field recording
- Limited duplication and distribution of audio programs
- Production of metal parts (stampers) for the record pressing plant
The use of electric transcription disc recording outlived wire recording and may even prove to outlive magnetic tape recording. High quality lacquer transcription discs are still used today as part of the process of mastering vinyl LPs and singles.
With limited resources, decisions on which discs to preserve can be difficult. We try to offer here some guidelines for selecting among professionally recorded discs (known content) as well as home recordings (possibly unknown content).
Most professional recordings were labeled with at least some notations written on them. On the other hand, these labels were also prone to falling off as the label glue dried with time.
For the moment, lets assume that you know the content of your recordings and that you have a sense of which ones are most important from a content point of view.
ALL transcription discs are at risk of loss due to deterioration, without exception. When evaluating discs for risk of loss, use the following guidelines. We order the discs from highest priority to lowest priority for preservation based on present condition:
- Delaminating discs. It may be too late to save the audio contained in the delaminated portion of the disc. The delaminating process cannot be halted, so these make the top of the list. Delamination can be observed most frequently at the edges of the disc, but can also start in the middle of the disc by bubbling. The bubbles can be very small creating an almost stippled look, or the bubbles can be quite large (an inch or two in size). With extreme care, skill and some luck, the audio can still be saved on the remaining adhered and contiguous grooves.
- Cracked laminate. This is one step away from delaminating. Individual pieces of laminate can easily unadhere from the substrate and peel off. These discs may not even be playable anymore if the cracks are too wide. They will already be difficult to play as the stylus will be prone to skipping and repeating depending on the orientation of the crack(s). You may have one or two plays remaining in these discs before they delaminate.
- Glass substrate discs. They are so fragile and accident prone - and so difficult to repair when damaged that even if the disc looks good, it needs to be preserved sooner than later.
- Palmitic and stearic acid deposits. These appear as a white waxy/greasy coating, and in the early stages look like a fine white dust. This is an early stage of deterioration where the plasticizer has started exuding from the laminate causing the laminate to shrink and become more brittle. This will eventually lead to delamination. Fortunately, the acid deposits can be removed when using the right chemistry and equipment and a good transfer can often be made.

This disc is severely delaminated with missing pieces. Amazingly, the grooved area was intact and complete near the center of the disc. This disc was preserved just in time.

The laminate on this disc (a very early original John Cage recording) has swelled and delaminated. You can see that this 16-inch disc is fully supported using a custom support platter. After much hard work, we successfully transferred all the grooves on this disc.

Example of cracked laminate that is not initially obvious because the cracks follow the grooves. More often, cracks in the lamination tend to be radial.

An advanced case of palmitic acid coating on a disc. Note how there is no palmitic acid formation under the cellophane tape used to secure the disc label. The tape sealed the area under it from moisture, preventing the formation of palmitic acid.
Selecting Among Unknown Content
When dealing with home recordings, people are often faced with a pile of unlabeled discs with no idea of their content. The discs may have rare recordings of family members, or may simply have dubs of favorite songs or radio programs. The cost of preserving all the discs may not be practical or within their budgets, and there is probably no way to preview the discs to figure out what is on them.
How do you select the few discs to be transfered among many discs of unknown content?
Look for the most played discs - the discs with the most scratches and gouges - because these were the most played. And they were most played because their content was probably the most interesting or entertaining.
Unfortuantely, because these discs were most played, damaged and worn, they will also be the most compromised (ie. noise) when transferred. Nonetheless, this method of disc selection tends to work well when determining the recordings with the most personal or cultural value when presented with a large number of discs from which to make selections.
The good news is that we have many professional digital restoration tools that can be used to make your well-worn (we prefer "well loved") recordings sound as good as possible.
Construction and Materials of Transcription Discs
A transcription disc consists of two major components:
- Substrate
- Laminate
The substrate is a base material that forms the disc and which is then coated (laminated) with a softer material in which the audio grooves are cut. Substrate materials include:
- Aluminum (most common)
- Steel (World War II era discs)
- Glass (World War II era discs)
- Fiber (cardboard-like)
Aluminum and steel substrate discs are mechanically sturdy. However, they are susceptible to permanent bending, warping and other deformities if mis-handled. Nonetheless, these discs must be kept flat as bends and warps make them difficult to play, and because they are metal, the bends and warps are difficult to remove.
Glass substrate discs are incredibly fragile and need to be handled with extreme care. IMPORTANT: To the untrained eye, they look just like aluminum substrate discs. Their fragility cannot be over-emphasized. Glass discs are not easily identified. Some ways to identify glass discs:
Glass discs most often appear in collections that were recorded between 1941 and 1945. Glass discs were used during World War II in order to conserve aluminum. After World War II there was still unused inventory of glass discs, which means that glass discs can appear in collections recorded after World War II. We have encountered glass disc recordings made as late as 1948 and later.
Glass discs originally came in envelopes that identified them as glass. If you are lucky, they will still be in their original envelopes. Often times the envelopes get switched around, so you cannot count on glass discs to be labeled.
Glass discs are a little heavier than aluminum discs, but the weight difference is subtle and unless you've handled glass discs with regularity, you won't be able to tell the difference between them and aluminum.
Glass discs sound different than aluminum when gently tapped. Once again, the difference is subtle and not obvious unless you have experience.
Glass discs can sometimes be identified by looking at the inside of the spindle hole or the other holes. If the inside of the hole doesn't look shiny and silver, or like fiber/cardboard - the disc is probably glass. IMPORTANT: This test does not guarantee that the disc is NOT glass - read on...
Glass discs were also made as a glass anulus (ring) with a small aluminum or fiber disc at the center which contains the spindle and other holes. When looking inside the holes, you will think you have an aluminum or fiber-based disc when in fact you might have a glass disc. However, in this case there often is a visible seam or ridge between the inner aluminum or fiber disc and the surrounding glass. This seam may be obscured by the label.
Glass discs can sometimes be identified by looking at the edge of the disc. Glass discs might have a rougher edge (but not always!). If the substrate edge is exposed due to the laminate being imperfect or chipped, it might be possible to identify if the substrate is metal or glass. Do not remove substrate at the edge of the disc in an attempt to identify the substrate - this can become a point of future deterioration of the disc.
Fiber substrate discs are not as fragile as glass, but still must be handled with great care because they bend and flex easily. Bending and flexing of the disc will cause the laminate (coating with the grooves) to crack or even peel. They are easy to identify because they are lighter than metal and sound like thin cardboard or thick paper when lightly tapped. Because the fiber easily abosrbs moisture from the air, these discs will expand and shrink over time and with the seasons if not stored in an environmentally controlled space (constant humidity). This constant shrinking and expanding of the substrate will often cause the laminate to crack and craze, giving it an "alligatored" appearance.
The laminate is the coating into which the grooves are cut. Prior to the use of nitrocellulose (also known as Cellulose Nitrate) was the use of soft wax, ethyl cellulose, and cellulose acetate. In fact the misnomer "acetate" derives from the brief period where discs were coated with cellulose acetate. Unless you have transcription disc recorded before 1934, in all likelihood the laminate is nitrocellulose.
The challenge in creating a laminate for cutting grooves is that it needs to be soft enough for cutting, yet hard enough to be played back more than once. Transcription discs can in fact be played more than once, but the grooves are worn down a little with each play. Even just a half-dozen plays under ideal circumstances will create distinctly audible wear.
The use of the correct stylus size will help reduce the wear on the one hand, but if the correct stylus is determined by trial and error by playing the disc - this process puts additional wear on the grooves. The Audio Archive has developed processes to minimize groove wear when selecting styli.
In order to make the nitrocellulose layer softer for cutting grooves, a plasticizer was added to the coating. The plasticizer was castor oil.
Next we will discuss the mechanical and chemical properties of the nitrocellulose laminate in the context of handling, cleaning and storage of transcription discs.
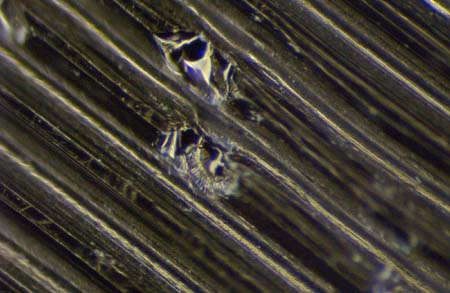
This is what happens when a stylus is dropped onto the soft surface of a transcription disc. The two gouges are where the stylus tore through the groove wall on either side of a groove, and was audible in all three adjacent grooves. These gouges were photographed through our microscope at 150x.
Repeat: DO NOT TOUCH THE SURFACE of a transcription disc without gloves.
Finger prints are acidic, and palmitic acid will form where there are finger prints. The palmitic acid will form precisely on the finger print with perfect detail, and over time will etch the surface and grooves of the laminate. Even after thoroughly cleaning the disc and removing all palmitic and stearic acid, the etched finger print will be both visible and audible (as noise).
It cannot be emphasized enough that transcription discs always be handled with gloves.
Supporting a 16-inch disc edge-to-edge throughout the cleaning and transfer process (especially with glass) minimizes any substrate deflection, and hence minimizes laminate or substrate cracking (glass). We have special platters that we use to handle glass transcription discs - we move a disc sandwiched between two of these special platters to prevent the possibility of breakage. These platters also serve as supports for cleaning and playback, which further minimizes handling of the transcription disc. In addition to handling, these support platters are very useful for working with broken discs. The broken disc can be assembled on the support platter, and later when sandwiched between platters, the re-assembled pieces can also be easily flipped to play the opposite disc side.
Cleaning of Electric Transcription Discs
Be forewarned, this is a rather dense and long discussion, but cleaning of transcription discs is a critical element in their transfer and preservation.
Transcription discs are prone to developing a coating of a white waxy or greasy substance, which in its early stages looks like a fine white dust or powder. This substance is palmitic acid and stearic acid. Many in the archival community simply refer to this as palmitic acid or "PA" for short.
The early "dust" or powder stage of palmitic acid formation is sometimes mistaken for mold, but under the microscope one can clearly distinguish whether it is mold or palmitic acid.

An example of a moderate coating of palmitic acid on a 16-inch disc. You can actually read the word "audiodisc" in the palmitic acid. It appears that the original sleeve (paper) allowed less moisture to pass through the paper where the audiodisc logo was printed on the sleeve - so less palmitic acid formed under the logo. Our custom 16-inch platter supporting the disc on the Keith Monks record cleaning machine is visible.
A clear understanding of palmitic acid and its formation are essential to cleaning transcription discs.
So, what is palmitic acid?
The best way we have found to think of and explain palmitic acid, is that it is a kind of "margarine". Palmitic acid (and stearic acid, the other part of that white greasy film that you find on transcription discs) is basically hydrogenated castor oil, castor oil being the plasticizer in the cellulose nitrate. Continuing with the margarine analogy, margarine is basically hydrogenated vegetable oil. Both palmitic acid and margarine share carboxylic acid (for example, palmitic acid and stearic acid) in common. You can read more about this in the ARSC journal article: "Selected Acetate Disc Cleaning Methods" (1997, Paton et al). A brief quote: "...we propose that the castor oil used as a plasticizer reacted with water vapor in the air (humidity). In the presence of water vapor, one or more of the ester bonds can hydrolyze, releasing the various carboxylic acids. This process is accelerated when acids are present and the PH of the acqueous environment is below 7."
Although the "margarine" analogy is one that we came up with, the hydrolyzing of the castor oil by breaking one or more ester bonds, releasing various carboxylic acids comes directly from the aforementioned ARSC journal article.
Keeping with the margarine analogy, if you are cleaning any fatty acid, what might you use? If you look at a restaurant, they reach for the clear ammonia. We recommend this same ingredient as part of a cleaning solution - NH4OH (also known as ammonium hydroxide, or "clear ammonia") available on most supermarket shelves. Some advantages of clear ammonia are that it is: (1) affordable and (2) has no additional ingredients/additives like other commercial cleaners containing ammonia (for example, window cleaners).
We mix a 1-2% clear ammonia solution with our record cleaning solution to create our transcription disc cleaning solution. A typical formulation might be:
- 50 parts Disc Doctor
- 50 parts distilled deionized water
- 1-2 parts clear ammonia
The clear ammonia will rapidly cut through the palmitic and stearic acid deposits and leave you with an absolutely clean transcription disc afterwards when used in conjunction with a record cleaning solution like Disc Doctor. In fact, the use of clear ammonia finds indirect support in the same 1997 ARSC Journal Article, except that the authors of that article found that Kodak Lens Cleaner worked best for cleaning palmitic acid. But if you take a close look at the active ingredient in Kodak Lens Cleaner you find none other than (NH4)2CO3 (ammonium carbonate) - a close cousin to NH4OH (ammonium hydroxide).
The chemistry of the cleaning process with palmitic acid is pretty clear. The palmitic acid is an "acid", of course , and ammonia hydroxide is a "base" which neutralizes and breaks down the acid.
As a final cleaning step we inspect the bottom of the groove with a 150x microscope, and especially check the corners between the groove bottom and the walls for any remaining palmitic and stearic acid deposits. You can learn to recognize palmitic acid residue in the groove bottoms with the naked eye once you familiarize yourself with what it looks like through a microscope. But the microscope is best for positive verification. In some cases, a worn groove can be mistaken for palmitic acid residue with the naked eye. So having a microscope available is important for ensuring a clean disc as well as preventing over-cleaning of the disc grooves.
Palmitic acid can also develop into a crystalline form which is particularly difficult to clean. In this case we suggest a slightly higher concentration of clear ammonia (3-4%):
- 50 parts Disc Doctor
- 50 parts distilled deionized water
- 3-4 parts clear ammonia
We recommend distilled deionized water rather than just distilled water. Deionized water has additional scavenging properties because contaminants are often ionized, and therefore they are more attracted to the deionized water molecules.
One question that we often get is: whether to leave palmitic acid on a disc and wait to remove it until it is transferred or to remove it now?
Ideally, we would recommend cleaning the disc now , removing the palmitic acid deposits, and rehousing the disc in a base-buffered sleeve. We recommend this approach because:
- palmitic acid deposits are part of an autocatalytic reaction - the deterioration process accelerates in the presence of acid, which then generates more acid, which further accelerates deterioration, in an endless spiral.
- palmitic acid deposits will etch the groove walls over time, making the recording more noisy. It does not require much palmitic acid to etch the groove wall in the nitrocellulose laminate. Leaving palmitic acid on the discs will cause imprinting to the disc surface (a kind of roughness due to accelerated exuding of plasticizer and hence laminate shrinkage beneath the palmitic acid) which then shows up as noise in future transfers.
This needs to be balanced against the reality of the cost of cleaning and rehousing a transcription disc collection, and also against the risk of damage associated with additional handling of this fragile media. At a minimum, if possible, triage your collection for the discs with the worst palmitic acid and just clean and rehouse those. However, do NOT clean discs that are delaminating or where the lamination is bubbling or cracking.
Palmitic acid formation varies, and here are some patterns that we have observed. These are not hard and fast rules, but when triaging a collection for cleaning of palmitic acid, these guideline might be helpful:
- Home recordings generally have less palmitic acid formations. This is because home recordings had less plasticizer in the laminate in order to make them harder and somewhat more durable for multiple plays.
- Glass transcription discs seem to have less palmitic acid formations in comparison to metal substrate discs stored under the same conditions. We hypothesize that this is because glass is an insulator and therefore responds to changes in temperature more slowly, causing less plasticizer to be exuded from the laminate.
- Fiber substrate discs seem to have less palmitic acid formations both because these tend to be home recordings, but also because the fiber does not respond to temperature changes as rapidly as the metal substrate discs.
Cleaning methods we do NOT advocate:
- Lighter fluid
- Mineral oil
We have not found any scientific explanation or evidence that these other cleaning methods (lighter fluid, mineral oil) work, and for this reason we take the conservative position of not advocating their use.
Some further discussion and clarification of cleaning process details is warranted.
The primary considerations when selecting a cleaning process for transcription discs are the following:
- Contact time with water must be minimized because the cellulose nitrate laminate is hygroscopic, meaning it absorbs water. When the laminate abosrbs water, it swells and can even bubble and delaminate. The swelling is NOT reversible - this means that if the moisture in the laminate is removed, the deformation of the laminate will not change (it will remain swollen and bubbled).
- Mechanical scrubbing must be minimized because the cellulose nitrate laminate is soft and can be damaged by excessive scrubbing.
These two factors determine the cleaning process choices and details.
For example, we have had great success with Disc Doctor cleaning solutions and brushes for disc cleaning in general (vinyl, shellac, and lacquer), and highly recommend these products, however we feel that for palmitic acid deposits, a modified approach is required by adding ammonium hydroxide. Disc Doctor, on the other hand, advocates using a highly concentrated version of the Disc Doctor cleaning solution for removing palmitic acid deposits rather than adding ammonium hydroxide. We have tested non-diluted Disc Doctor cleaning solution, and have found that far too much mechanical scrubbing and time are required to break down and remove the palmitic acid when dealing with heavy coatings of palmtic acid. By simply adding 1-4 parts of clear ammonia to the Disc Doctor cleaning solution, we can reduce the time to break down and remove the palmitic acid by a factor of 4x. This dramatically reduces contact time with water and reduces mechanical scrubbing of the disc.
The second critical element of cleaning transcription discs is rapidly removing the cleaning and rinse solutions from the disc surface. We prefer NOT to just air dry transcription discs after rinsing . Rather, the cleaning and rinse solutions should be removed using a vacuum-style record cleaning machine which sucks the water from the surface of the disc. We use and recommend the Keith Monks Archivist Uno Record Cleaning Machine Mk IV.

Kieth Monks record cleaning machine with custom 16-inch support platter and a fluid splash fence surrounding the machine. The Keith Monks RCM rests on a stainless steel shelf for easy cleaning. All brushes and cleaning solutions are on shelves at platter height for easy access and efficient workflow. A high intensity light is above the cleaning machine to highlight contamination.
Some important notes about the Keith Monks record cleaning machine specifically and record cleaning machines in general:
- When cleaning 16-inch discs, the cleaning or rinse fluid will quickly find its way into the cleaning arm bearing, which can rust in as little as 24 hours. We highly recommend:
- Shielding the bearing from fluid
- Spraying WD-40 into the bearing to displace any water should water get into the cleaning arm bearing
- NOTE: this is not a problem with 12-inch discs cleaned on the 16-inch Keith Monks Archivist Uno RCM Mk IV.
- The Disc Doctor cleaning brushes are even more effective than the Keith Monks built-in brush. We would love it if Keith Monks made a lower-cost version of the Archivist Uno without the brush to make this marvelous machine more affordable.
- Extreme care must be taken with 16-inch discs cleaned on the Keith Monks record cleaning machine which has a 12-inch support platter. This means that two inches of the perimeter of the disc are unsupported. 16-inch glass transcription discs can easily be cracked. The Audio Archive uses custom 16-inch support platters to ensure that the disc is never exposed to bending forces.
- Be generous with cleaning and rinse fluids. This may seem counter-intuitive because water is not good for transcription discs. But the cleaning and rinse solutions work more effectively when the disc is flooded, which ultimately minimizes fluid contact time when used in conjunction with a Keith Monks or similar record cleaning machine.
- Multiple cleaning and rinse passes may be required to remove all the palmitic acid. Usually this is the case with heavy palmitic acid deposits or crystalline palmitic acid formations. This again underscores the importance of minimizing fluid contact time if multiple passes are required to break down all the palmitic acid.

A baby bottle nipple with its top cut off is used to shield the cleaning arm bearing against water entry
The Audio Archive has developed a process for microscopically examining and accurately meauring groove shape and dimensions. We then compute the ideal stylus size for an individual disc side through the use of somewhat complicated geometric calculations that we developed, tested and verified.
We make the initial stylus selection without ever playing the record or putting wear on the grooves. We examine the grooves using a 150x microscope that has a calibrated measurement reticle capable of 5-10 micron resolution. Because we have examined the grooves at a microscopic level, we can already anticipate problems of groove wear and cutting problems, and modify our stylus selection accordingly - and we still haven't touched the groove with a stylus.
The Audio Archive has helped leading archives around the USA adopt this method of stylus selection by assisting them with microscope selection and set-up, and training them on its use and how to make the calculations.
In general, the stylus used for transcription discs will range in size from just under 2.0 mils to maybe just over 3.0 mils. Much depends on the quality of the transcription disc blank and thickness of the laminate, the skill of the recording engineer, and the condition of the cutting stylus. Home recordings will have the most variability in groove dimensions.
It is also worth noting that just because Side A of a transcription uses a 2.5 mil stylus doesn't mean that Side B will also. Recording engineers will sometimes make adjustments to the cutting parameters between sides, and a microscope will catch these changes.
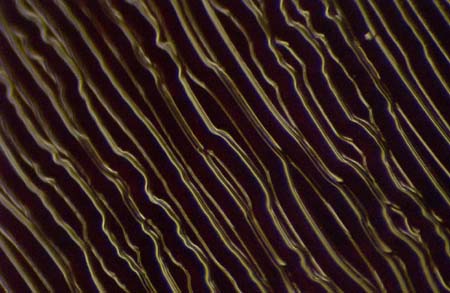
Grooves are cut so close to each other that groove walls are touching, resulting in cross-talk. Problems like this can be anticipated using a microscope before ever even putting a stylus in the groove. Photographed on our microscope at 150x.
Assuming that you have the proper stylus and the transcription disc is not deteriorating (delaminating, bubbling or cracking), the disc can then be played without any special techniques. Naturally, a high quality turntable is required with appropriate armboard, tonearm and platter if the discs are 16-inch.
Playback of deteriorated transcription discs require a well set-up turntable (good antiskate settings especially) in order to track through any cracks in the laminate. The tonearm that we use is the SME 312 (about $2000) that allows us to play discs in poor condition - even broken discs or discs with missing bits of laminate in some cases.
Another useful tool is a 2-inch horse hair paint brush. This will allow you to apply very gentle anti-skate and vertical forces selectively during playback in order to track through difficult grooves.
A solid understanding of the relationship between VTF (Vertical Tracking Force) and anti-skate is an essential piece of knowledge for the transfer engineer to have. We cover this in the tutorials and talks we have given at the AES and ARSC Conferences, and we also review this as part of the training courses that we give to clients.

State-of-the-art playback is achieved for all discs using a Simon Yorke S7 archival turntable with dual-armboard and SME tonearms. A Nikon 150x microscope with a 5-10 micron resolution graduated reticle and fiber optic lighting is mounted over the platter. The turntable rests on a Minus K atomic force microscope isolator to eliminate any external vibration to the turntable.
There are three bad actors that affect the long-term condition of transcription discs when stored:
- Temperature variation - causes plasticizer to exude which then causes the nitrocellulose laminate to shrink and eventually delaminate. In addition, the exuded plasticizer starts the cycle of palmitic acid formation and the long-term damage to the groove walls.
- Moisture - breaks down the ester bonds in the castor oil plasticizer forming palmitic and stearic acid
- Acidity - accelerates the formation of palmitic and stearic acid. This is an autocatalytic reaction which is hard to stop once started
Hence the storage of transcription discs should comprise:
- Environmental control is critical for storage (low and stable temperature and low relative humidity (RH)).
- Acid-free (base buffered) sleeves for housing discs after cleaning enhances disc longevity by staving off palmitic acid formation.
Following is a more detailed analysis of transcription disc deterioration and storage requirements.
The macro environment is most important - stable and low temperature and low humidity - in order to minimize the exuding of castor oil plasticizer from the disc and the ensuing formation of palmitic and stearic acid.
But what about the micro environment? What sort of housing would be best for transcriptoin discs?
Plasticizer loss is unavoidable over time, so the next level of protection is (a) lack of water and (b) a non-acidic micro environment to slow formation of carboxylic acids (palmitic and stearic acid).
The nitrocellulose itself can create an acidic micro environment, therefore local air circulation and a well-buffered container will be important. Pickett and Lemcoe, 1959, provides some support for this: "The most signficant decomposition product [of cellulose nitrate] is nitrogen peroxide which is converted into nitrous or nitric acid in the presence of liquid water and, since an acid has a catalytic effect on the degradative reactions, this is an autocatalytic reaction." So once again, the primary movers in the degradation process are (a) water and (b) acids formed by the reaction with water.
Based on the Pickett and Lemcoe research, as well as the research done by Paton et al, this simply reinforces the importance of having a dry environment for nitrocellulose discs to minimize the formation of acidic compounds from the decomposition of the nitrocellulose (forming nitric acid) and the castor oil (forming carboxylic acids). Because a water-free environment is impossible to create, and may have undesirable side effects, the formation of nitric acid and palmitic and stearic acids over time are unavoidable.
But the autocatalytic effects that accelerate degradation can be minimized through the use of a properly buffered, acid- and lignin-free sleeve. Air circulation can help dissipate acidic vapors, but since you cannot necessarily count on air circulation in a record stack, the higher pH buffer will be the first line of defense against the acid formations.
Based on this conclusion, polyethylene inner sleeves are not appropriate for nitrocellulose transcription discs because they lack any buffer properties.
Finally, discs should be stored vertically. When stored horizontally, the pressure on the discs at the bottom of the stack will have impressions of the sleeve or the disc above it pressed into the laminate, potentially damaging the groove walls and the audio. We prefer to store transcription discs in base buffered sleeves that are then stored in base buffered boxes for maximum protection against palmitic acid.
Based on the above analysis, we recommend the following products from Bags Unlimited:
- Bags Unlimited S16LP (16-inch disc sleeve, base buffered)
- Bags Unlimited S12WHD (12-inch disc, acid free, not base buffered)
- Bags Unlimited S10LP (10-inch disc sleeve, base buffered)
- Bags Unlijmited S7WHD (7-inch disc, acid free, not base buffered)
- Bags Unlimited XLP16BB (16-inch disc box, base buffered)
- Bags Unlimited XLP12BB (12-inch disc box, base buffered)
Although the 12-inch and 7-inch sleeves are acid free, they are not base buffered, which limits their ability to neutralize acids that form on the disc. By using these sleeves in conjunction with the barrier board boxes (which are base buffered), this will provide some protection against an acidic environment from building up.
Resources
Disc Doctor
We recommend the Disc Doctor Miracle Record Cleaner solutions and wet brushes
Kieth Monks
Our record cleaning machine of choice is the Keith Monks Archivist Uno RCM Mk. IV
Bags Unlimited
The 16-inch disc sleeves from Bags Unlimited provide excellent value and preservation performance. See our list of specific sleeve product codes.